 |
| Willi Hennig four years before his death |
Specifically,
the taxonomy of the lower Brachycera is now an exemplar of phylogenetic
instability. These flies were once classified as a taxon called Orthorrhapha,
but recognition that this group simply constituted all brachycerans that did not
belong in the Cyclorrhapha put an end to its usage by
dipterists; the orthorrhaphous superfamily Empidoidea is obviously sister to
the Cyclorrhapha*, and together they constitute the Eremoneura. Beyond that,
the phylogeny becomes (for lack of a better word) hairy: the Asiloidea may be
either eremoneurans' adelphotaxon, or they may be paraphyletic—with
the Bombyliidae and Mythicomyiidae their own clade (Bombylioidea), and/or with Apystomyia
elinguis (translating more or less to "the fly of which nothing is
known"; Marshall, 2012) constituting its own family and classified as the
Eremoneura's sibling (Wiegmann et al., 2011).
 |
| An adult wormlion (probably Lampromyia sp.), photographed by Valter Jacinto |
The Nemestrinoidea, another group of lower Brachycera, are in a similar pickle. This (putative) superfamily contains two most peculiar taxa (Woodley, 1989): the Nemestrinidae and Acroceridae (formerly Cyrtidae). Their broader relationships are debatable, as is their relation to each other (Woodley et al., 2009): despite the many ecological parallels between the two, which will now be detailed.
 |
| A Virginian Ogcodes borealis, noticed by Scott Justis |
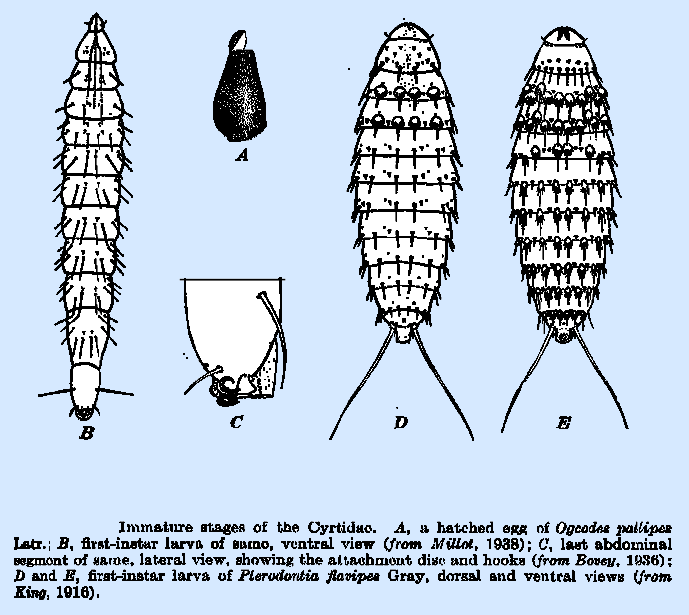 Additionally,
all acrocerid larvae seek out their hosts autonomously upon hatching (usually
nocturnally; Cole, 1919); the hyperactive first instar is termed a planidium:
the description and ontological purpose of which will be recalled by anyone who
is familiar with mantidflies (with which nemestrinoids converge markedly).
Rapid and broad dispersal is imperative; a number of acrocerid planidia bear a
posterior duo of elongated setae (as seen at left)—these facilitate leaping
(Cole, 1919; Bovey, 1936). Planidia of Acrocera, despite belonging to
the type genus of the family, deviate from the norm by affixing themselves to a
given substrate and awaiting some unfortunate spider's passage
(Schlinger, 2003).
Additionally,
all acrocerid larvae seek out their hosts autonomously upon hatching (usually
nocturnally; Cole, 1919); the hyperactive first instar is termed a planidium:
the description and ontological purpose of which will be recalled by anyone who
is familiar with mantidflies (with which nemestrinoids converge markedly).
Rapid and broad dispersal is imperative; a number of acrocerid planidia bear a
posterior duo of elongated setae (as seen at left)—these facilitate leaping
(Cole, 1919; Bovey, 1936). Planidia of Acrocera, despite belonging to
the type genus of the family, deviate from the norm by affixing themselves to a
given substrate and awaiting some unfortunate spider's passage
(Schlinger, 2003).  |
| A female Ogcodes eugonatus ovipositing on a clothesline; photographed by J. T. Layne in Missouri |
 |
| A collected S. appendiculata, male, in lateral view |
 |
| Moegistorhynchus longirostris sipping from an iris (Lapeirousia anceps) (Zhang et al., 2012) |
 |
| Atriadops sp., female, on an Australian windowscreen; photographed by Alexander Pierce |
 |
| Hirmoneura flavipes, caught in Arizona by Margarethe Brummermann |
Trichopsideines and nemestrinines plague grasshoppers (Acrididae) (Prescott, 1955) (the host[s] of Cyclopsidea are so far undiscovered). Planidia penetrate the host's integument where it is softest, adjacent to the abdominal spiracles; after wandering the tracheae for a few days, they cut a different hole in the trachea (in Trichopsidea clausa; York & Prescott, 1952) or directly on the host's exoskeleton near a spiracle (in T. flavopilosa and the nemestrinine Neorhynchocephalus) and press their rearmost spiracles to the newly-made pore, subsequently burrowing deep into the grasshopper's entrails: and a ropy umbilicus then gradually forms from the host tissue, maintaining the larva's connection to the orifice it previously chewed, thereby permitting respiration. Nemestrinids sometimes take these respiratory tubes to proportions unseen among other parasitoids that utilize the same principle—in T. flavopilosa and costata (Potgieter, 1929), the final-instar larva's tube is twice its bodily length. If and when the host moults, the larva must create an entirely new pore, or else suffocate (Prescott, 1961).
 |
| Wing of Neorhynchocephalus sp., pictured by Robert Behrstock and exhibitng typically nemestrinid venation |
The fact that the earliest undoubted fossil of a small-headed fly only makes an appearance around 100 million years after the oldest known tangle-veined one (Mostovski, 1997; Grimaldi et al., 2002; Shi et al., 2012) could be a distinct strike against these insects' monophyly: chronological dissociation helped lead to the separation of the Mythicomyiidae from the Bombyliidae (Evenhuis, 2012). And thus are we led to the subject of nemestrinoid phylogeny. For many years, the Nemestrinoidea were regarded as closely akin to bee flies (Bombyliidae) merely by dint of the parasitoidal ontogeny they have in common (Hennig, 1973; Ovtshinnikova, 1998); but this treatment has been abandoned in favor of either situating the Nemestrinoidea near the Asiloidea (Woodley, 1989) or within the Tabanomorpha (Griffiths, 1994). Increasingly, Acroceridae and Nemestrinidae are phyletically dissociated, since they lack any unassailable synapomorphies (Woodley et al., 2009; Wiegmann et al., 2011).
 |
| A teneral stink fly (Coenomyia ferruginea) from Wisconsin, photographed by Marcie O'Connor |
In any event, Sinonemestrius appears to provide us with a morphological link between the Xylophagidae and Nemestrinidae (Jarzembowski & Mostovski, 2000), at least. The Acroceridae, however, remain regrettably ambiguous.
Kudos to anyone who recognizes the literary reference made in this post's title...
*An unranked clade, sometimes synonymized with the infraorder Muscomorpha. More accurately, that infraorder refers to the Cyclorrhapha and the former Asilomorpha. In any event, cyclorrhaphous flies are united by the circularity of the opening they make in their puparium upon eclosion.
†Almost entirely consisting of eyes.
‡One of two basal lobes in a fly's wing, absent in a goodly number of species.
______________________________________________________________
Ansorge, J. and Mostovski, M. (2000). Redescription of Prohirmoneura jurassica Handlirsch, 1906 (Diptera, Nemestrinidae) from the Lower Tithonian lithographic limestone of Eichstätt (Bavaria). Neues Jahrbuch für Geologie und Paläontologie Monatshefte, 4, 235-243.
Bequaert, J. C. and Carpenter, F. M. (1936). The Nemestrinidae of the Miocene of Florissant, Colorado, and their relations to the recent fauna. Journal of Palaeontology, 10, 395-409.
Borkent, C. J. and Schlinger, E. I. (2008). Pollen loads and pollen diversity on Eulonchus tristis (Diptera: Acroceridae): implications for pollination and flower visitation [electronic version]. The Canadian Entomologist, 140(2), 257-264. Retrieved 9/22/13 from http://journals.cambridge.org/action/displayAbstract?fromPage=online&aid=8484583
Barraclough, D. and Slotow, R. (2010). The South African pollinator Moegistorhynchus longirostris (Wiedemann, 1819) (Diptera: Nemestrinidae): notes on biology, biogeography and proboscis length variation [electronic version]. African Invertebrates, 51(2), 397-403. Retrieved 9/23/13 from http://www.researchgate.net/publication/232691797_The_South_African_Keystone_Pollinator_Moegistorhynchus_longirostris_%28Wiedemann_1819%29_%28Diptera_Nemestrinidae%29_Notes_on_Biology_Biogeography_and_Proboscis_Length_Variation
Bernardi, N. (1973). The genera of the family Nemestrinidae (Diptera, Brachycera). Arquivos de Zoologia, 24(4), 211-318.
Bovey, P. (1936). Sur la ponte et la larvae primare d'Ogcodes pallipes. Latreille Soc. Vaud. des Sci. Nat. Bul., 59, 171-176.
Brauer, F. (1883). Erganzende Bemerkungen zu a. Handlirsch's Mittheilungen über Hirmoneura obscura. Mg. Wien. Ent. Stg., 2, 25-26.
Cady, A.; Leech, R.; Sorkin, L.; Stratton, G.; and Caldwell, M. (1993). Acrocerid (Insecta: Diptera) life histories, behaviors, host spiders (Arachnida: Araneida), and distribution records. The Canadian Entomologist, 125, 931-944.
Clausen, C. P. (1940). Entomophagous Insects. New York City: McGraw-Hill.
Cole, F. R. (1919). The dipterous family Cyrtidae in North America. Transcriptions of the American Entomological Society, 45, 1-69. Retrieved 9/14/13 from http://www.jstor.org/stable/pdfplus/25077002.pdf?acceptTC=true&
Coscarón, S.; Coscarón, M. C.; and Gil-Azevedo, L. H. (2013). On the enigmatic Heterostomus curvipalpis Bigot, 1857, with a description of the pupa (Diptera, Brachycera). Zootaxa, 3616(3), 268-276. Retrieved 9/30/13 from http://www.mapress.com/zootaxa/2013/f/z03616p276f.pdf
Devetak, D. (2008). Wormlions Vermileo vermileo (L.) (Diptera: Vermileonidae) in Slovenia and Croatia. Ann. Ser. Hist. Nat., 18(2), 283-286. Retrieved 9/2/13 from http://www.google.com/url?sa=t&rct=j&q=&esrc=s&source=web&cd=1&ved=0CDAQFjAA&url=http%3A%2F%2Fwww.dlib.si%2Fstream%2FURN%3ANBN%3ASI%3ADOC-HEQZ3FMC%2F574ad873-e8df-49f8-94e4-7c6da027d08b%2FPDF&ei=cBAlUtnjLsinsQTWsoCABQ&usg=AFQjCNE-8ef4xtP3jASFAkpQlIGhnPorkA&sig2=Avf9ZNXW2a5uDGLQ4m1mgw&bvm=bv.51495398,d.cWc
Evenhuis, N. L. (1994). Catalogue of the Fossil Flies of the World (Insecta: Diptera). Leiden: Backhuys.
Evenhuis, N. L. (2012). Family Mythicomyiidae. Catalog of the Diptera of the Australasian and Oceanian regions (online version). Retrieved 9/29/13 from http://hbs.bishopmuseum.org/aocat/aocathome.html
Gillung, J. P. and Winterton, S. L. (2011). New genera of philopotine spider flies (Diptera, Acroceridae) with a key to living and fossil genera. ZooKeys, 127(2011), 15-27. Retrieved 9/29/13 from http://www.pensoft.net/journals/zookeys/article/1824/abstract/new-genera-of-philopotine-spider-flies-diptera-acroceridae-with-a-key-to-living-and-fossil-genera
Grant, V. and Grant, K. A. (1965). Flower Pollination in the Phlox Family. New York City: Columbia University Press.
Griffiths,
G. C. D. (1994). Relationships among the major subgroups of Brachycera
(Diptera): a critical review. Canadian Entomology, 126, 861-880.
Grimaldi, D. A. (1999). The co-radiations of pollinating insects and angiosperms in the Cretaceous. Annals of the Missouri Botanical Garden, 86, 373-406.
Grimaldi, D. A.; Engel, M. S.; and Nascimbene, P. C. (2002). Fossiliferous Cretaceous amber from Myanmar (Burma): its rediscovery , biotic diversity, and paleontological significance. American Museum Novitates, 3361, 1-71.
Jarzembowski, E. A. and Mostovski, M. B. (2000). A new species of Sinonemestrius (Diptera: Brachycera) from the Weald Clay (Lower Cretaceous, southern England), with a discussion of its affinities and stratigraphical implications. Cretaceous Research, 21(2000), 761-765. Retrieved 9/30/13 from http://www.researchgate.net/publication/222656799_A_new_species_of_Sinonemestrius_%28Diptera_Brachycera%29_from_the_Weald_Clay_%28Lower_Cretaceous_southern_England%29_with_a_discussion_of_its_affinities_and_stratigraphical_implications?citationList=outgoing
Grimaldi, D. A. (1999). The co-radiations of pollinating insects and angiosperms in the Cretaceous. Annals of the Missouri Botanical Garden, 86, 373-406.
Grimaldi, D. A.; Engel, M. S.; and Nascimbene, P. C. (2002). Fossiliferous Cretaceous amber from Myanmar (Burma): its rediscovery , biotic diversity, and paleontological significance. American Museum Novitates, 3361, 1-71.
Jarzembowski, E. A. and Mostovski, M. B. (2000). A new species of Sinonemestrius (Diptera: Brachycera) from the Weald Clay (Lower Cretaceous, southern England), with a discussion of its affinities and stratigraphical implications. Cretaceous Research, 21(2000), 761-765. Retrieved 9/30/13 from http://www.researchgate.net/publication/222656799_A_new_species_of_Sinonemestrius_%28Diptera_Brachycera%29_from_the_Weald_Clay_%28Lower_Cretaceous_southern_England%29_with_a_discussion_of_its_affinities_and_stratigraphical_implications?citationList=outgoing
Johnson,
S. D. (2010). The pollination niche and its role in the diversification and
maintenance of the southern African flora. Philosophical Transactions of the
Royal Society Series B, Biological Sciences; 365, 499-516.
Kanmyia, K. (1987). New records of endoparasitic Nemestrinidae and Tachinidae (Diptera) on Tettigoniidae. Acta Dipterologica, 15, 14.
Karolyi, F.; Szucsich, N. U.; Colville, J. F.; and Krenn, H. W. (2012). Adaptations for nectar-feeding in the mouthparts of long-proboscid flies. Biological Journal of the Linnaean Society, 107, 414-424. Retrieved 9/23/13 from http://onlinelibrary.wiley.com/doi/10.1111/j.1095-8312.2012.01945.x/pdf
Kanmyia, K. (1987). New records of endoparasitic Nemestrinidae and Tachinidae (Diptera) on Tettigoniidae. Acta Dipterologica, 15, 14.
Karolyi, F.; Szucsich, N. U.; Colville, J. F.; and Krenn, H. W. (2012). Adaptations for nectar-feeding in the mouthparts of long-proboscid flies. Biological Journal of the Linnaean Society, 107, 414-424. Retrieved 9/23/13 from http://onlinelibrary.wiley.com/doi/10.1111/j.1095-8312.2012.01945.x/pdf
Kerr, P. H. and Winterton, S. L. (2008). Do parasitic flies attack mites? Evidence in Baltic amber [electronic version]. Biological Journal of the Linnaean Society, 93(1), 9-13. Retrieved 9/16/13 from http://era.deedi.qld.gov.au/1203/
King, J. L. (1916). Observations on the life history of Pterodontia flavipes Gray [electronic version]. Annals of the Entomological Society of America, 9, 309-321. Retrieved 9/18/13 from http://www.ingentaconnect.com/content/esa/aesa/1916/00000009/00000003/art00005
Larrivée, M. and Borkent, C. J. (2009). New spider host associations for three acrocerid fly species [electronic version]. Journal of Arachnology, 37(2), 241-242. Retrieved 9/26/13 from http://www.bioone.org/doi/abs/10.1636/T08-62.1
Ludwig, P.; Melzer, R. R.; and Ehrhardt, V. (2001). Larval morphology and classification of wormlions (Diptera: Vermileonidae). Mitt. Dtsch. Ges. Allg. Angew. Ent., 13, 89-94.
Marshall, S. (2012). Flies: the Natural History and Diversity of Diptera. Richmond Hill: Firefly Publishing.
Montgomery, T. H. (1903). Studies on the habits of spiders, particularly those of the mating period. Proceedings of the National Academy of Sciences of Philadelphia, 65, 58-149.
Mostovski, M. B. (1997). To the knowledge of fossil dipterans of superfamily Archisargoidea (Diptera, Brachycera). Palaeontological Journal, 1, 72-77.
Mostovski, M. B. (1998). A revision of the Nemestrinid flies (Diptera, Nemestrinidae) described by Rohdendorf, and a description of new taxa of the Nemestrinidae from the Upper Jurassic of Kazakhstan. Palaeontological Journal, 32, 369-375.
Mostovski, M.B. and Martínez-Delclòs, X. (2000). New Nemestrinoidea (Diptera: Brachycera) from the Upper Jurassic-Lower Cretaceous of Eurasia, taxonomy and paleontology [electronic version]. Entomological Problems, 31(2), 137-148. Retrieved 9/30/13 from http://fossilinsects.net/pdfs/Most_Delclos_2000LR.pdf
Nagatomi, A. (1977). Classification of lower Brachycera (Diptera). Journal of Natural History, 11, 321-335.
Nagatomi, A. (1992). Notes on the phylogeny of various taxa of the orthrrhaphous Brachycera (Insecta: Diptera). Zoological Sciences, 9, 843-857.
Nagatomi, A. and Yang, D. (1998). A review of extinct Mesozoic genera and families of Brachycera (Insecta, Diptera, Orthorrhapha). Entomologist's Monthly Magazine, 134, 95-192.
Narchuk, E. P. (2007). Nemestrinid flies (Diptera, Nemestrinidae) in the fauna of Eastern Europe and the Caucasus [electronic version]. Retrieved 9/29/13 from http://download.springer.com/static/pdf/15/art%253A10.1134%252FS0013873807080143.pdf?auth66=1380654571_05e4f8df1ad77ac2fdac6f7dd646fec0&ext=.pdf
Nel, A. (2010). A new Mesozoic-aged rhagionemestriid fly (Diptera: Nemestrinoidea) from China. Zootaxa, 2645, 49-54. Retrieved 9/30/12 from http://mapress.com/zootaxa/2010/f/z02645p054f.pdf
Nielsen, B. O.; Funch, P.; and Toft, S. (1999). Self-injection of a dipteran parasitoid into a spider. Naturwissenschaften, 86(11), 530-532. Retrieved 9/12/13 from http://www.ncbi.nlm.nih.gov/pubmed/10551947
Ovtshinnikova, O. G. (1998). A brief review of male genital musculatures in Brachycera Orthorrhapha (Insecta, Diptera) with special reference to phylogenetic relationships of families. Proceedings of the Zoological Institute of RAS, 276, 143-147.
Paramonov, S. J. (1955). New Zealand Cyrtidae (Diptera) and the problem of the Pacific island fauna. Pacific Science, 9, 16-25.
Pauw, A.; Stofberg, J.; and Waterman, R. J. (2009). Flies and flowers in Darwin's race. Evolution, 63, 268-279.
Potgieter, J. T. (1929). A contribution to the biology of the brown swarm locust Locusta pardalina (Wlk.) and its natural enemies. Pan-African Agricultural and Veterinary Conference, Pretoria; Proceedings of the Agricultural Section, 265-238.
Potgieter, C. J.; Edwards, T. J.; Miller, R. M.; and van Staden, J. (1999). Pollination of seven Plectranthus spp. (Lamiaceae) in southern Natal, South Africa. Plant Systematics and Evolution, 218, 99-112.
Prescott, H. W. (1955). Neorhynchocephalus sackenii and Trichopsidea clausa: nemestrinid parasites of grasshoppers [electronic version]. Annals of the Entomological Society of America, 48(5), 392-402. Retrieved 9/28/13 from http://www.m3cg.us/hopper/content/neorhynchocephalus-sackenii-and-trichopsidea-clausa-nemestrinid-parasites-grasshoppers
Prescott, H. W. (1961). Respiratory pore construction in the host by the Nemestrinid parasite Neorhynchocephalus sackenii (Diptera), with notes on respiratory tube characters. Annals of the Entomological Society of America, 54(4), 557-566. Retrieved 9/29/13 from http://www.ingentaconnect.com/content/esa/aesa/1961/00000054/00000004/art00021?crawler=true
Richter, V. A. (1997). Family Nemestrinidae. In Papp, L. and Darvas, B. (eds.) (pp. 459-468): Contributions to a Manual of Palaearctic Diptera, vol. 2. Budapest: Science Herald.
Riley, C. V. (1883). Larval stages and habits of the bee-fly Hirmoneura. Science, 1, 332-334.
Schlinger, E. I. (1981). Acroceridae. In McAlpine, J. F.; Peterson, B. V.; Shewell, G. E.; Teskey, H. J.; Vockeroth, J. R.; and Wood, D. E. M. (eds.) (pp. 575-584): Manual of Nearctic Diptera, vol. 1. Agriculture Canada: Research Branch.
Schlinger, E. I. (1987). The biology of Acroceridae (Diptera): true endoparasitoids of spiders. In Nentwig, W. (ed.) (pp. 319-327): Ecophysiology of Spiders. Berlin: Springer Verlag.
Schlinger,
E. I. (2003). Acroceridae, spider-fly endoparasitoids. In S. M. Goodman and J.
P. Bernstead (eds.) (pp. 734-740): The Natural History of Madagascar. Chicago:
University of Chicago Press.
Schlinger,
E. I.; Gillung, J. P.; and Borkent, C. J. (2013). New spider flies from the
Neotropical region (Diptera, Acroceridae) with a key to New World genera. ZooKeys,
270(2013), 59-93. Retrieved 9/19/13 from http://www.pensoft.net/J_FILES/1/articles/4476/4476-G-1-layout.htm
Sferra,
N. J. (1986). 1st record of Pterodontia flavipes (Diptera, Acroceridae)
larvae in the mites Podothrombium (Acari, Trombidiidae) and Abrolophus
(Acari, Erythraeidae) [electronic version]. Entomological News, 97, 121-123.
Retrieved 9/16/13 from http://biostor.org/reference/77015
Shi, G.; Grimaldi, D. A.; Harlow, G. E.; Wang, J.; Wang, J.; Yang, M.; Lei, W.; Li, Q.; and Li, X. (2012). Age constraint on Burmese amber based on U-Pb dating of zircons. Cretaceous Research, 37(2012), 155-163. Retrieved 9/29/13 from http://www.sciencedirect.com/science/article/pii/S0195667112000535?np=y
Stuardo, C. (1935). Algunas observaciones sobre las costumbres y metamorfosis de Hirmoneura articulata. Ph. Rev. Chilena de Hist. Nat., 1934, 197-202.
Triplehorn, C. A. and Johnson, N. F. (2005). Borror and DeLong's Introduction to the Study of Insects (7th ed.). Belmont: Thomson Brooks/Cole.
Ussatchov, D. A. (1968). New Jurassic Asilomorpha (Diptera) of the fauna of Karatau. Entomological Review, 47(3), 617-628.
Wedmann, S. (2007). A nemestrinid fly (Insecta: Diptera: Nemestrinidae: cf. Hirmoneura) from the Eocene Messel Pit (Germany) [electronic version]. Journal of Paleontology, 81(5), 1,114-1,117. Retrieved 9/29/13 from http://www.jstor.org/stable/4498863?seq=3
Woodley, N. E.; Borkent, A.; and Wheeler, T. A. (2009). Phylogeny of the Diptera. In Brown, B. V.; Borkent, A.; Cumming, J. M.; Wood, D. M.; Woodley, N. E.; Zumbado, M. A. (eds.) (pp. 9-50): Manual of Central American Diptera, vol. 1. Ottawa: NRC Research Press.
Yeates, D. K. (1994). The cladistics and classification of Bombyliidae (Diptera: Asiloidea) [electronic version]. Bulletin of the American Museum of Natural History, 219, 1-191. Retrieved 9/24/13 from http://digitallibrary.amnh.org/dspace/handle/2246/826
York, G. T. and Prescott, H. W. (1952). Nemestrinid parasites of grasshoppers. Journal of Economic Entomology, 45, 5-10.
Zhang, F.; Hui, C.; and Pauw, A. (2012). Adaptive divergence in Darwin's race: how coevolution can generate trait diversity in a pollination system [electronic version]. Evolution, 67(2), 548-560. Retrieved 9/22/13 from http://onlinelibrary.wiley.com/doi/10.1111/j.1558-5646.2012.01796.x/pdf
Shi, G.; Grimaldi, D. A.; Harlow, G. E.; Wang, J.; Wang, J.; Yang, M.; Lei, W.; Li, Q.; and Li, X. (2012). Age constraint on Burmese amber based on U-Pb dating of zircons. Cretaceous Research, 37(2012), 155-163. Retrieved 9/29/13 from http://www.sciencedirect.com/science/article/pii/S0195667112000535?np=y
Stuardo, C. (1935). Algunas observaciones sobre las costumbres y metamorfosis de Hirmoneura articulata. Ph. Rev. Chilena de Hist. Nat., 1934, 197-202.
Triplehorn, C. A. and Johnson, N. F. (2005). Borror and DeLong's Introduction to the Study of Insects (7th ed.). Belmont: Thomson Brooks/Cole.
Ussatchov, D. A. (1968). New Jurassic Asilomorpha (Diptera) of the fauna of Karatau. Entomological Review, 47(3), 617-628.
Wedmann, S. (2007). A nemestrinid fly (Insecta: Diptera: Nemestrinidae: cf. Hirmoneura) from the Eocene Messel Pit (Germany) [electronic version]. Journal of Paleontology, 81(5), 1,114-1,117. Retrieved 9/29/13 from http://www.jstor.org/stable/4498863?seq=3
Wiedemann,
C. R. W. (1830). Familie der Feistfliegen (Inflatae). Aussereuropaische
Zweiflügelege Insekten, 2, 13-20.
Woodley, N. E. (1989). Phylogeny and classification of the "Orthorrhaphous" Brachycera. In McAlpine, J. F. (ed.) (pp. 1371-1395): Manual of Nearctic Diptera, vol. 3. Agriculture Canada: Research Branch.
Wiegmann,
B. M.; Trautwein, M. D.; Winkler, I. S.; Barr, N. B.; Kim, J.; Lambkin, C.;
Bertone, M. A.; Cassel, B. K.; Bayless, K. M.; Heimberg, A. M.; Wheeler, B. M.;
Peterson, K. J.; Pape, T.; Sinclair, B. J.; Skevington, J. S.; Blagoderov, V.;
Caravas, J; Kutty, S. N.; Schmidt-Ott, U.; Kampmeier, G. E.; Thompson, F. C.;
Grimaldi, D. A.; Beckenbach, A. T.; Courtney, G. W.; Friedrich, M.; Meier, R.;
and Yeates, D. K. (2011). Episodic radiations in the fly tree of life. Proceedings
of the National Academy of Sciences, United States; 108.
Winterton,
S. L.; Wiegmann, B. M.; and Schlinger, E. I. (2007). Phylogeny and Bayesian
divergence time estimations of small-headed flies (Diptera: Acroceridae) using
multiple molecular markers [electronic version]. Molecular Phylogenetics
& Evolution, 43(3), 808-832. Retrieved 9/27/13 from http://www.sciencedirect.com/science/article/pii/S105579030600323X
Winterton,
S. L. and Gillung, J. P. (2012). A new species of spider fly in the genus Sabroskya
Schlinger from Malawi, with a key to Acrocerinae world genera. ZooKeys,
171(2012): 1-15. Retrieved 9/5/13 from http://www.pensoft.net/journals/zookeys/article/2137/abstract/a-new-species-of-spider-fly-in-the-genus-sabroskya-schlinger-from-malawi-with-a-key-to-acrocerinae-world-genera-diptera-
Woodley, N. E. (1989). Phylogeny and classification of the "Orthorrhaphous" Brachycera. In McAlpine, J. F. (ed.) (pp. 1371-1395): Manual of Nearctic Diptera, vol. 3. Agriculture Canada: Research Branch.
Woodley, N. E.; Borkent, A.; and Wheeler, T. A. (2009). Phylogeny of the Diptera. In Brown, B. V.; Borkent, A.; Cumming, J. M.; Wood, D. M.; Woodley, N. E.; Zumbado, M. A. (eds.) (pp. 9-50): Manual of Central American Diptera, vol. 1. Ottawa: NRC Research Press.
Yeates, D. K. (1994). The cladistics and classification of Bombyliidae (Diptera: Asiloidea) [electronic version]. Bulletin of the American Museum of Natural History, 219, 1-191. Retrieved 9/24/13 from http://digitallibrary.amnh.org/dspace/handle/2246/826
York, G. T. and Prescott, H. W. (1952). Nemestrinid parasites of grasshoppers. Journal of Economic Entomology, 45, 5-10.
Zhang, F.; Hui, C.; and Pauw, A. (2012). Adaptive divergence in Darwin's race: how coevolution can generate trait diversity in a pollination system [electronic version]. Evolution, 67(2), 548-560. Retrieved 9/22/13 from http://onlinelibrary.wiley.com/doi/10.1111/j.1558-5646.2012.01796.x/pdf