 |
| Male Panorpa nuptialis (Panorpidae) photographed by Gayle and Jeanell Strickland |
—this being based on molecular and morphological lines of evidence (Wood & Borkent, 1989; Whiting, 2002; Simiczyjew, 2002; Beutel & Baum, 2008); although this conclusion is still open to debate (Wiegmann et al., 2009; Beutel et al., 2010; Friedrich et al., 2012).
 |
| Ventral view of female Caurinus tlagu (Boreidae: Caurininae) (Sikes & Stockbridge, 2013) |
 |
| Ventral view of male Merope tuber captured in New Hampshire by Jace Porter; forceps are readily visible |
Earwigflies are not distinctive for this alone: what with their flattened profile, ornate wing venation, opisthognathous‡ head, and castaneous coloration the family is utterly unlike any sympatric mecopteran. These features correlate with earwigflies' nocturnal and ground-dwelling habits—unobtrusive customs that have foiled attempts to study their biology; the fact that adults stridulate by rasping the jugum§ against thoracic serrations (Sanborne, 1982; Dunford et al., 2006) is really the only detail we have gleaned, and the larvae have never been documented (Johnson, 1995).
 |
| Notiothauma reedi (Eomeropidae), photographed in Chile by David Madison |
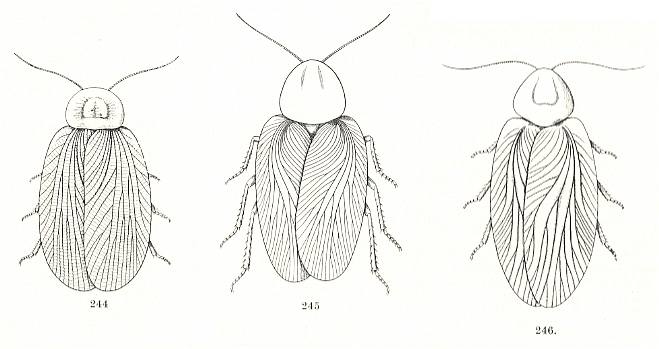 |
| Archimylacrids (the two at left) and spiloblattinid (right); image in public domain (I hope) |
Holistic consideration of eomeropid characteristics is suggestive of that family's inclusion within the clade Pistillifera (all Mecoptera exclusive of Boreidae and Nannochoristidae), since the perceived similarities that united the protomecopteran taxa were plesiomorphies (Mickoleit, 1971; Willmann, 1981; Friedrich et al., 2012). This leaves earwigflies hanging loose: the Meropeidae may either be nested well within the Pistillifera, the sister-group to the remainder of that clade (Friedrich et al., 2012), or perhaps even the basalmost twig of their order (Penny, 1975). In all likelihood, meropeid phylogeny will remain opaque until more data is acquired—anything concerning the ever-elusive larva would be especially useful.
Well, that's over with. Now to walk through the Valley of the Shadow of Calculus...
*Scorpions' (Scorpiones) "stinger".
†Heavily compressed, sclerotized, and lacking abdominal prolegs, as in the larvae of Elateridae (click beetles).
‡Rearward-oriented (with reference to mouthparts).
§A basal posterior lobe of an insect forewing.
______________________________________________________________
Albornoz, S.; Ortloff, A.; de la Fuente, J. C.; and Vivallo, G. (2015). Notiothauma reedi, a new species of forensic importance to the forests of southern Chile. Oral presentation. Retrieved 5/31/15 from http://www.researchgate.net/publication/271193099_Notiothauma_reedi_especie_forense_en_bosques_de_Chile
Archibald, S. B.; Rasnitsyn, A. P.; and Akhmetiev, M. A. (2005). Ecology and Distribution of Cenozoic Eomeropidae (Mecoptera), and a New Species of Eomerope Cockerell from the Early Eocene McAbee Locality, British Columbia, Canada. Annals of the Entomological Society of America, 98(4), 503-514. Retrieved 6/6/15 from http://www.brucearchibald.com/docs/Archibald_et_al_2005.pdf
Beutel, R. G. and Baum, E. (2008). A longstanding entomological problem finally solved? Head morphology of Nannochorista (Mecoptera, Insecta). Journal of Zoological Systematics and Evolutionary Research, 46(4), 346-347. Retrieved 2/24/15 from http://www.researchgate.net/publication/229883320_A_longstanding_entomological_problem_finally_solved_Head_morphology_of_Nannochorista_%28Mecoptera_Insecta%29_and_possible_phylogenetic_implications
Beutel, R. G.; Friedrich, F.; and Whiting, M. F. (2008). Head morphology of Caurinus (Boreidae, Mecoptera) and its phylogenetic implications. Arthropod Structure & Development, 37, 418-433.
Beutel, R. G.; Zimmermann, D.; Krauß, M.; Randolf, S.; and Wipfler, B. (2010). Head morphology of Osmylus fulvicephalus (Osmylidae, Neuroptera) and its phylogenetic implications. Organism's Diversity & Evolution, 10, 311-329.
Byers, G. W. (1965). New and uncommon Neotropical Mecoptera. Journal of the Kansas Entomological Society, 38(2), 135-144. Retrieved 5/31/15 from http://www.jstor.org/stable/25083427?seq=1#page_scan_tab_contents
Byers, G. W. (1973). Zoogeography of the Meropeidae (Mecoptera). Journal of the Kansas Entomological Society, 46(4), 511-516.
Crampton, G. C. (1930). The wings of the remarkable archaic mecopteran Notiothauma reedi McLachlan with remarks on their Protoblattoid affinities. Psyche, 37(1), 83-103.
Dunford, J. C.; Serrano, D.; and Somma, L. A. (2006). Earwigflies in the Great Smokies. Southeast Biology, 53(1), 27-29. Retrieved 5/31/15 from http://www.fsca-dpi.org/SEBiolEarwigfly.pdf
Fraulob, M.; Wipfler, B.; Hünefeld, F.; Pohl, H.; and Beutel, R. G. (2012). The larval abdomen of the enigmatic Nannochoristidae (Mecoptera, Insecta). Arthropod Structure & Development, 41(2), 187-198. Retrieved 2/24/15 from http://www.sciencedirect.com/science/article/pii/S1467803911001149
Friedrich, F.; Pohl, H.; Beckmann, F.; Beutel, R. G. (2012). The head of Merope tuber (Meropeidae) and the phylogeny of Mecoptera (Hexapoda). Arthropod Structure & Development, 42(1), 69-88. Retrieved 2/24/15 from http://www.sciencedirect.com/science/article/pii/S1467803912000771
Grimaldi, D. and Engel, M. S. (2005). Evolution of the Insects. Cambridge: Cambridge University Press.
Grimaldi, D. A. and Engel, M. S. (2013). The relict scorpionfly family Meropeidae (Mecoptera) in Cretaceous amber. Journal of the Kansas Entomological Society, 86, 253-263. Retrieved 6/6/15 from http://www.bioone.org/doi/abs/10.2317/JKES130219.1?af=R
Hepburn, H. R. (1969). The proventriculus of Mecoptera. Journal of the Georgia Entomological Society, 4, 159-167.
Hinton, H. E. (1981). Biology of Insect Eggs. New York: Pergamon Press.
Johnson, N. F. (1995). Variation in male genitalia of Merope tuber Newman (Mecoptera: Meropeidae). Journal of the Kansas Entomological Society, 68(2), 224-233.
Kristensen, N. P. (1991). Phylogeny of extant hexapods. In Naumann, I. D.; Cornell, P. B. C.; Lawrence, J. F.; Neilson, E. S.; Spradberry, J. P.; Taylor, R. W.; Whitten, M. J.; and Littlejohn, M. J. (eds.): The Insects of Australia: a Textbook for Students and Research Workers (2nd ed.) (pp. 125-140). CSIRO, Melbourne: Melbourne University Press.
Liu, S.; Shih, C. K.; and Ren, D. (2014). Four new species of hangingflies (Insecta, Mecoptera, Bittacidae) from the Middle Jurassic of northeastern China. ZooKeys, 466, 77-94. Retrieved 2/24/15 from http://zookeys.pensoft.net/articles.php?id=4370
Machado, R. J. P.; Kawada, R.; and Rafael, A. J. (2013). New continental record and new species of Austromerope (Mecoptera, Meropeidae) from Brazil. ZooKeys, 269, 51-65. Retrieved 5/30/14 from http://zookeys.pensoft.net/articles.php?id=3699&display_type=element&element_type=4&element_id=417&element_name=
Mickoleit, G. (1971). Zur phylogenetischen und funktionellen Bedeutung der sogenannten Notalorgane der Mecoptera (Insecta: Mecoptera). Zeitschrift für Morphologie der Tiere, 83, 483-496.
Novokshonov, V. (1995). Der älteste Vertreter der Meropeidae (Mecoptera, Insecta). Palaeontologische Zeitschrift, 69, 149-152.
Peña, L. E. (1968). Natural history notes on Notiothauma. Discovery, 4, 42-44.
Penny, N. D. (1975). Evolution of the extant Mecoptera. Journal of the Kansas Entomological Society, 48(3), 331-350.
Rasnitsyn, A. P. (1974). Taxonomic names, in New Mesozoic and Cenozoic Protomecoptera. Paleontological Journal, 8, 493-507.
Rebolledo, R. R.; Alda, L. L.; Gutiérrez, M. S.; and Guiñez, B. S. (1990). Antecedentes biológicos de Notiothauma reedi MacLachlan (Mecoptera: Eomeropidae) en el Monumento Natural Cerro Ñielol (Temuco, Chile). Revista Chilena de Entomología, 18, 25-28.
Riek, E. F. (1953). Fossil mecopteroid insects from the Upper Permian of New South Wales. Records of the Australian Museum, 23, 55-87.
Sanborne, P. M. (1982). Stridulation in Merope tuber (Mecoptera: Meropeidae). The Canadian Entomologist, 114(3), 177-180.
Sikes, D. S. and Stockbridge, J. (2013). Description of Caurinus tlagu, new species, from Prince of Wales Island, Alaska (Mecoptera, Boreidae, Caurininae). ZooKeys, 316, 35-53. Retrieved 2/24/15 from http://zookeys.pensoft.net/articles.php?id=3342
Simiczyjew, B. (2002). Structure of the ovary in Nannochorista neotropica Navás (Insecta: Mecoptera: Nannochoristidae) with remarks on mecopteran phylogeny. Acta Zoologica, 83, 61-66.
Somma, L. A. and Dunford, J. C. (2007). Etymology of the earwigfly, Merope tuber Newman (Mecoptera: Meropeidae): simply dull or just inscrutable? Insecta Mundi, 13, 1-5.
Tillyard, R. J. (1935). The evolution of the scorpion-flies and their derivatives (Order Mecoptera). Annals of the Entomological Society of America, 28, 1-45.
Whiting, M. F. (2002). Mecoptera is paraphyletic: multiple genes and phylogeny of Mecoptera and Siphonaptera. Zoologica Scripta, 31(1), 93-104. Retrieved 2/24/15 from http://decapoda.nhm.org/pdfs/12600/12600.pdf
Wiegmann, B. M.; Trautwein, M. D.; Kim, J.-W.; Cassel, B. K.; Bertone, M. A.; Winterton, S. L.; and Yeates, D. K. (2009). Single-copy nuclear genes resolve the phylogeny of the holometabolous insects. BMC Biology, 7, 34. Retrieved 2/24/15 from http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2709105/
Willmann, R. (1981). Phylogenie und Verbreitungsgeschichte der Eomeropidae (Insecta: Mecoptera). Ein Beispiel für die Anwendung der phylogenetischen Systematik in der Paläontogie. Paläont. Z., 55, 31-49.
Wood, D. M. and Borkent, A. (1989). Phylogeny and classification of the Nematocera. In McAlpine, J. F. and Wood, D. M. (eds.): Manual of Nearctic Diptera, vol. 3 (pp. 1,333-1,370). Ottawa: Agriculture Canada.
Zhang, J.; Shih, C.; Petrulevičius, J. F.; and Ren, D. (2011). A new fossil eomeropid (Insecta, Mecoptera) from the Jiulongshan Formation, Inner Mongolia, China [electronic version]. Zoosystema, 33(4), 443-450. Retrieved 6/6/15 from http://www.bioone.org/doi/abs/10.5252/z2011n4a2?journalCode=zoos
No comments:
Post a Comment